 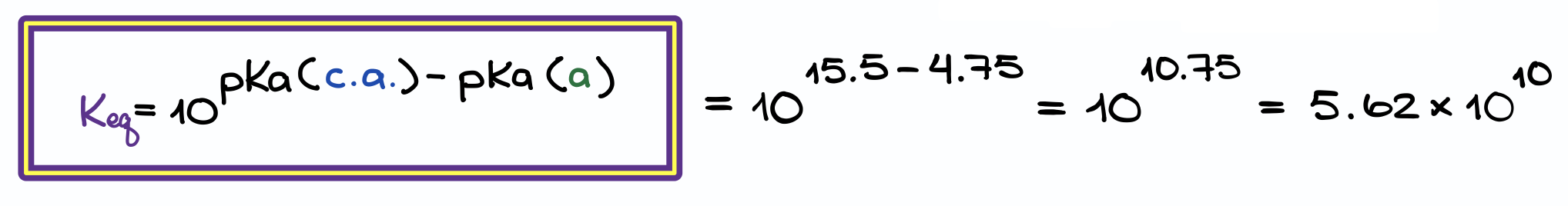
It’s impossible to sort out all the opposing variables this way. Trends are only qualitative guides to this topic. It’s extremely important, in fact.īut what do you do when you want to compare the acidity of two molecules that aren’t connected by a trend?įor instance, resonance increases the acidity of alcohols, but by how much? More than you’d increase acidity by changing OH to SH? What if you added an electron withdrawing group? The bottom line is that any factor which stabilizes the conjugate base will result in increased acidity. So last time we went through all the different trends that affect acidity. The Acidity Constant K a (And Its Negative Logarithm pK a ) Is An Extremely Useful Measure Of Acidity 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
